Get Involved in Medical Research: Take Part, Raise Awareness

At the start of this year, I delightedly joined the National Institute for Health Research (NIHR) as a patient research ambassador (PRA) for the dermatology and musculoskeletal departments at The Royal Free Hospital, London.
According to their website, the NIHR is “the most integrated clinical research system in the world. [They] drive research from bench to bedside for the benefit of patients and the economy.” The NIHR funds high-quality research, trains and supports researchers, and provides world-class facilities, among other things.
This role is a huge honor for me, and I am VERY passionate about it. Here’s a video about my experience as a PRA.
I am also very passionate about my NHS England clinical reference group role and the European Medicines Agency (EMA) role.
All of these roles influence the medical research landscape in ways ranging from supporting the design of a clinical trial (via my EMA role) to shaping best practice guidelines (via my NHS England role).
More recently, I was the “Page 3 Girl” in my local newspaper, The Southport Champion, published in June, which was Scleroderma Awareness Month. I am truly honored and humbled to have been quoted on the front page of the recently published NIHR annual report. I was also featured later in the report.
I was overjoyed that the NIHR annual report was shared by various global media, including Med-Tech Innovation News and European Pharmaceutical Manufacturer quoting me, and naming Raynaud’s and scleroderma in their headline story.
These features put the spotlight on Raynaud’s and scleroderma, and the huge significance of medical research within the rare disease community. Rare disease patients know all too well that any and every day is an opportunity to engage conversation about our reality.
Medical research is the crux to uncovering the cause and cure to not only scleroderma and Raynaud’s, but any disease.
This is why I have made medical research the underlying theme for all pieces I have written for my column with Scleroderma News and my personal blog during the last few years.
Side note: Medical research has been the theme for Rare Disease Day in both 2017 and 2018.
During my 21 years of living with diffuse scleroderma and Raynaud’s, I have taken part in several clinical trials. All of these trials were led by Prof. Chris Denton at The Royal Free Hospital’s Scleroderma Unit.
It was very important for me to know that such an expert team led the trials. I am not the most willing of participants for taking any medication, licensed or unlicensed, even in the best of times. However, I saw my participation as an opportunity to investigate if there was a mystery compound that could improve my day-to-day reality with my symptoms. Even if I don’t benefit directly from these clinical trials, my hope is that patients diagnosed in the future may benefit from the therapy if the trials are a success.
Sadly, one of the difficulties of researching a rare disease is the lack of eligible patients available for clinical trials.
This is why global collaboration, as well as global databases and expert specialist centers, are paramount to making progress in the medical research arena for the rare disease community.
My patient profile campaigns for my Scleroderma Awareness Month 2017 and 2018 campaigns showed that for optimal patient care, three hallmarks preside:
- Early diagnosis
- Expert specialist centers
- Access to innovative medicines
Investment in medical research is crucial to improving quality of life and the long-term outlook for the entire global scleroderma community.
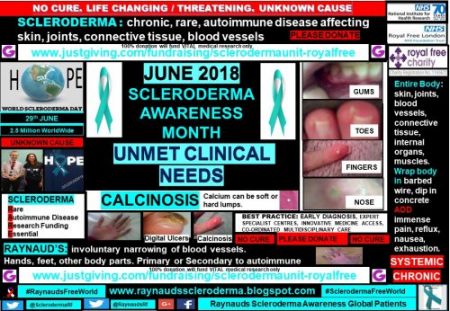
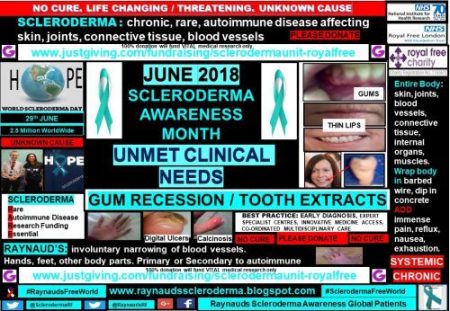
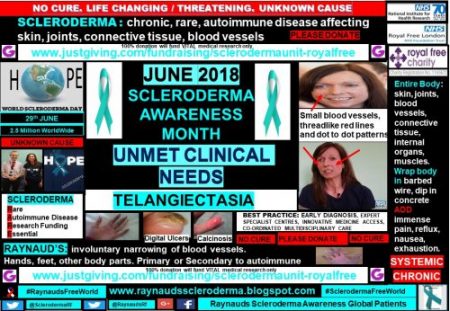
Although scleroderma patients are relatively few, 2.5 million worldwide, the small sample of patients included within my global patient profile campaign show that there are many unmet clinical needs.
Calcinosis
Gum recession and tooth extractions
Soft tissue ulcers
Telangiectasia:
Sclerodactyly hands
Invisible disability
Raynaud’s phenomenon
Gastrointestinal
Although these unmet needs are not immediately life-threatening, they cause havoc in each patient’s life quality. Investment in medical research will most certainly improve the currently soul-destroying reality these unmet clinical needs bring.
Living the dream, scleroderma style.
Follow:
Facebook Page: Raynauds Scleroderma Awareness Global Patients
Twitter: @SclerodermaRF @RaynaudsRf
Google Plus: RaynaudsSclerodermaAwarenessGlobalPatients
#SclerodermaFreeWorld #RaynaudsFreeWorld
Please DONATE to help fund medical research at The Scleroderma Unit, The Royal Free Hospital, London.
100 percent of raised funds will be used for medical research purposes only. Thank you.
#IAMHOPE #IAMSCIENCE
***
Note: Scleroderma News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website. The opinions expressed in this column are not those of Scleroderma News or its parent company, Bionews Services, and are intended to spark discussion about issues pertaining to scleroderma.





















Leave a comment
Fill in the required fields to post. Your email address will not be published.