Scleroderma Patient in Long-Term Remission with TPE Treatment
A new case study revealed that long-term treatment with therapeutic plasma exchange (TPE) led to successful symptom management and disease remission in a patient with CREST syndrome, or limited systemic scleroderma. The study, recently presented at the AABB Annual Meeting in California, is entitled “Benefit of Long-Term Therapeutic Plasma Exchange Treatment in a Patient with CREST Syndrome (Limited Systemic Scleroderma): A 21-Year Success Story,” and its abstract was published in the journal Transfusion.
CREST syndrome belongs to the heterogeneous group of sclerodermas, rare autoimmune diseases that affect the skin and potentially also several organs. CREST stands for the clinical features of the disease, namely calcinosis (formation of calcium deposits), Raynaud phenomenon (reduced blood flow in response to cold or emotional stress), esophageal dysmotility (impairment in esophagus contractions), sclerodactyl (localized thickening and tightness of the skin of the fingers or toes) and telangiectasia (dilatation of the capillaries, also called spider veins).
Current treatments for scleroderma conditions are based on immunosuppressants to avoid disease progression, and on interventions to help patients cope with specific symptoms.
The case report presents a male patient with CREST syndrome, diagnosed in early 1990 at age 43. After diagnosis, the patient developed additional symptoms, including severe Raynaud’s, a decline in pulmonary function, cardiac arrhythmias, and GERD (gastroesophageal reflux disease) with erosive esophagitis (inflammation and ulceration of the esophageal lining).
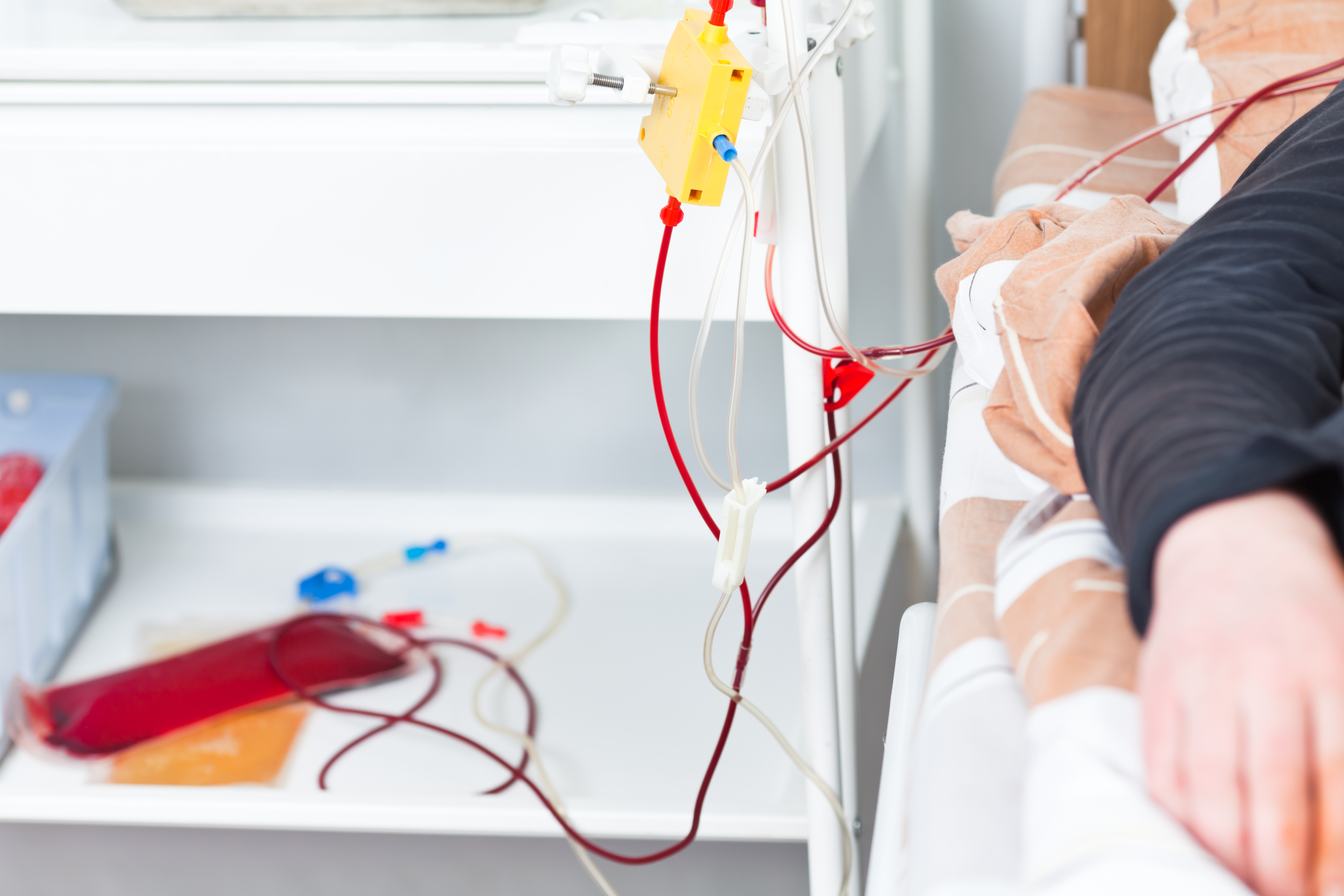
The patient started treatment in 1993 through regular therapeutic plasma exchange (TPE, also referred to as plasmapheresis), an extracorporeal technique in which potential blood-circulating harmful factors, like antibodies, are removed from the blood. The treatment was performed once a week for four weeks followed by two months of rest, totaling 16 TPE treatments per year (with the exception of one six-month suspension). No medication was administered.
After one year of TPE treatment, researchers observed a significant improvement in the patient’s GERD and Raynaud’s symptoms, and stable lung function. Two years after the start of the treatment, GERD symptoms had completely resolved, Raynaud’s symptoms became very mild, and the patient’s lung function returned to normal levels. After four years, the patient no longer experienced cardiac arrhythmias.
In 1996, when the TPE treatment was temporarily suspended, researchers found the disease again progressed, with GERD symptoms reappearing.
The patient is continuing to receive regular TPE treatment, and according to the abstract presented, he is in excellent physical condition and without disease symptoms, the only exception being mild but persistent Raynaud’s.
This is the first study reporting the successful use of TPE for such a long period (21 years). As opposed to the steady progression of symptoms experienced by many patients with limited scleroderma, almost all the scleroderma-related symptoms reversed under regular TPE treatment after several years.
The authors concluded that TPE could be considered a treatment option for patients with limited scleroderma, and suggested that continuous treatment may be crucial to maintaining disease remission. Interestingly, the team proposes that TPE not only helps remove harmful factors from the blood but also disaggregates clumped red blood cells (hyperaggregation), which may be the trigger for the vascular damage and other disease-related symptoms in patients with scleroderma.
“To the best of my knowledge, there are no previous reports in the research literature of any patient with clearly diagnosed systemic scleroderma receiving a treatment that reversed all significant symptoms, such as impaired lung functioning and severe GERD, and has been able to remain in remission for more than 20 years with continued ongoing treatments,” Edward Harris, the corresponding author of the study, said in an exclusive interview to Scleroderma News, part of the Bionews Services group. “While regular plasmapheresis treatments are expensive and venous access can be an issue for some patients, any treatment that offers scleroderma patients the possibility of a symptom-free life needs to be considered as a treatment option for suitable patients.”
He added, “It is important to conduct full clinical trials to fully evaluate this treatment approach and, if found to be an effective treatment, determine the mechanism of actions in order to be able to develop easier and more cost-effective treatments in the future.”
Asked whether TPE could be considered an effective and feasible therapy for scleroderma patients based on this single study, Harris was understandably cautious. “A reasonable generalization from this case study and the other published literature on TPE and scleroderma is that it may be effective at the indicated frequency for patients with relatively early stage anticentromere positive limited scleroderma,” he said. “However, I do not feel that the results of the case study can be generalized to support the hypothesis that this frequency of TPE treatments would be equally effective with more aggressive diffuse variants of scleroderma.”
Diffuse scleroderma differs from limited scleroderma in being a rapidly progressing and more aggressive disease, and it can affect the skin and one or more internal organs, including the lungs, heart, kidneys, and esophagus.
Adverse effects in regular TPE treatment are usually not an issue, Harris told Scleroderma News. “Modern TPE has a very low rate of adverse effects (3%) and even these adverse events were mild. There are no known immunosuppressant problems with regular TPE at the frequency used in this case study (16 times a year). However, at significantly greater frequencies (e.g., weekly), long-term TPE can lead to immunosuppressant problems similar to what is seen when patients receive immunosuppressant treatments.”
The team is planning to submit the complete case report data for publication in a scientific journal soon. If you are interested in contacting the corresponding author of this case study, you can do so at [email protected]. Harris is the director of the Scleroderma Education Project, providing scleroderma patients and their families helpful and accurate information about the disease, and its diagnosis and treatment.






